
Oligonucleotide Testing Market Size and Forecast By Type (Instrument (Liquid Chromatography, Mass Chromatography, Capillary Electrophoresis), Software, Service) By Application (Research, Commercial) And Trend Analysis, 2014 - 2024
- Published: April, 2018
- Format: Electronic (PDF)
- Number of pages: 60
- Industry: Pharmaceuticals
Industry Insights
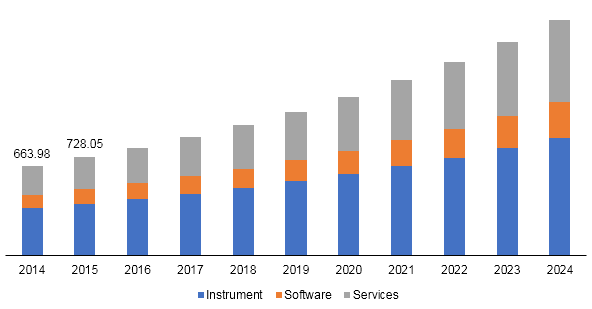
The global oligonucleotide testing market size was valued at USD 663.98 million in 2014 and is expected to witness a significant growth, pertaining to the rise in the technological advancements in synthesis of oligonucleotide products, introduction of strong pipeline programs, and increasing research activities in the segment over the forecast period. Oligonucleotide testing is majorly dependent on three factors namely modifications, application, and oligonucleotide length.
Oligonucleotide testing market revenue, by type, 2014 - 2024 (USD Million)
Various techniques are used in testing of oligonucleotide out of which Polyacrylamide gel electrophoresis (PAGE), Reverse Phase High Pressure Liquid Chromatography (RP HPLC), and Anion Exchange High Pressure Liquid Chromatography (AEX HPLC) are used majorly as these techniques provide highly accurate analytical screenings of oligonucleotide and provide optimum results. Technique such as IE-HPLC is used instead of RP-HPLC as IE-HPLC provides improved purification of nucleotides with substantial secondary structure, often found in sequences with higher GC content.
The pipeline includes more than 130 studies under clinical trials which consists of more than 30 companies. 37 out of the 137 are open studies and are recruiting. Remaining studies are either terminated or completed. Many of the completed studies are expected to show promising results. Majority of the clinical trials use siRNAs for treatment of different types of cancer. Recent approval of Spinraza (nusinersen) in December 2016 by the U.S. FDA has opened a new class of biologically derived products namely oligonucleotide drugs. Additionally, in January 2018, Spinraza was approved in Korea for treatment of Spinal muscular atrophy by the Korean Ministry of Food and Drug Safety. Such drug approvals in developing economies is expected to enhance market penetration of oligo testing in emerging economies over the forecast period.
Segmentation by type
• Instrument
• Liquid chromatography
• Mass chromatography
• Capillary electrophoresis
• Software
• Service
The instrument segment is expected to dominate the market and grow at a CAGR of 9.7% over the forecast period. Instrument segment is the major revenue generating segment and held a revenue share of more than 52% in 2016. High growth of this segment is majorly attributed to the introduction of advanced technology based chromatographic instruments. For instance, OST columns developed by Waters Corporation for the ultra-performance liquid chromatography (UPLC) analysis is suitable for efficient and faster separation of oligonucleotide in length up to 50 and longer.
Instruments such as LC-MS, UPLC, mass chromatography techniques such as PAGE and capillary electrophoresis are often used for analyzing and purity testing. While the drug molecule is a double chain DNA with complementary sequences, non-denaturing chromatographic or size exclusion chromatography techniques are also required. Multi angle light scattering and refractive index are commonly used for some conjugated oligonucleotide. In addition, LC-MS techniques are often used to establish the characteristic of oligonucleotide therapeutics and quantify impurities. Rising demand for synthetic gene on account of rapid increase in genetic disease is expected to booster growth. Owing to the growth in genetic disease has given importance to discover new advanced drugs to treat disease, which is further anticipated to upsurge the need for the oligonucleotide testing industry over the coming years.
Segmentation by application
• Research
• Commercial
The research segment is estimated to be fastest growing segment over the forecast period. The research segment held more than 67% of market share in 2016. The major share is attributed owing to the increased in R&D spending to develop effective oligonucleotide based drug and rising government initiatives. For instance, U.K Synthetic Biology is focusing on the processes of transformation and commercialization. About fifty start-ups, small and medium enterprises and larger companies are keenly focusing on synthetic biology in the U.K, and internal investments have started to flow.
Approximately, there were more than seventy oligonucleotide drugs in ongoing and/or recently completed clinical trials in the U.S. alone, which in turn is expected to augment growth in near future. Furthermore, increasing focus of pharmaceutical drug manufacturer on cost effective drugs is expected to propel the market over the forecast period. The market evolution is attributed to increasing number of pharmaceutical companies and the growing usage of oligonucleotide in research, therapeutics, and diagnostics.
Segmentation by region
• North America
• U.S.
• Europe
• Germany
• UK
• Asia Pacific
• China
• India
• Rest of the World (RoW)
In 2016, North America dominated the global market by contributing more than 40.2% and is expected to maintain its dominance owing to rising investment on R&D by biotechnology and pharmaceutical companies over the forecast period. The growth is derived by the presence of major players in the U.S. market coupled with the availability of private and public-sector funds for undertaking R&D activities in the region.
Europe is the second largest market followed by North America. Europe held the around 30% in 2016 and is expected to grow at a CAGR of nearly 8.8% over the forecast period. Increasing number of start-up companies coupled with adoption of new technologies is further contributing to the development of the market in the region. Additionally, rich patent portfolios along with the usage of newer technologies by pharmaceutical companies to build new molecules of oligonucleotides for research and therapeutic use is predicted to upsurge the market in Europe during the forthcoming years.
Competitive landscape
The global oligonucleotide testing market is fragmented in nature with a large number of existing and emerging new players. Major players in the industry comprise of companies such as Thermo Fisher Scientific, Integrated DNA Technologies (IDT), Merck KGaA, Agilent Technologies, Bruker and various others. Emerging players and new partnerships are driving the market. Competitors are taking strategic initiatives to increase their share. Presently, Thermo Fisher Scientific has a dominant and leading position owing to its strong product portfolio and robust outreach. Integrated DNA Technologies (IDT) and Merck KGaA (Sigma-Aldrich Corporation) capture significant market share.

Choose License Type
- World's largest premium report database
- Transparent pre & post sale customer engagement model
- Unparalleled flexibility in terms of rendering services
- Safe & secure web experience
- 24*5 Research support service
